HERV-Derived Shared Tumor Epitopes Identified by Complete Omics
A new class of shared, off-the-shelf immunotherapy targets derived from endogenous retroviruses was identified and validated with the support of Complete Omics’ Valid-NEO® platform.
Shared virus-like tumor antigens for next-generation cancer immunotherapy
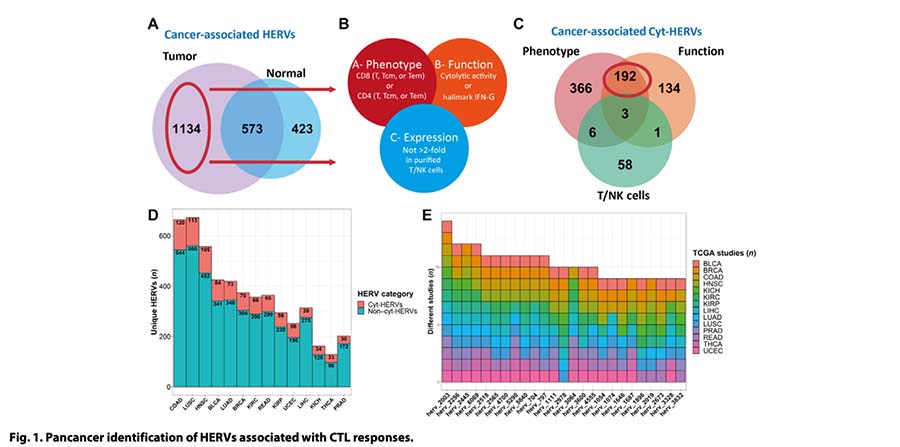
This landmark study demonstrates the power of our Valid-NEO® platform to unlock a completely new category of cancer immunotherapy targets: shared tumor epitopes derived from human endogenous retroviruses (HERVs). Unlike highly personalized neoantigens that vary from patient to patient, these HERV-derived targets are recurrent across tumors and therefore represent highly attractive candidates for scalable, off-the-shelf cancer vaccines and TCR-based therapies.
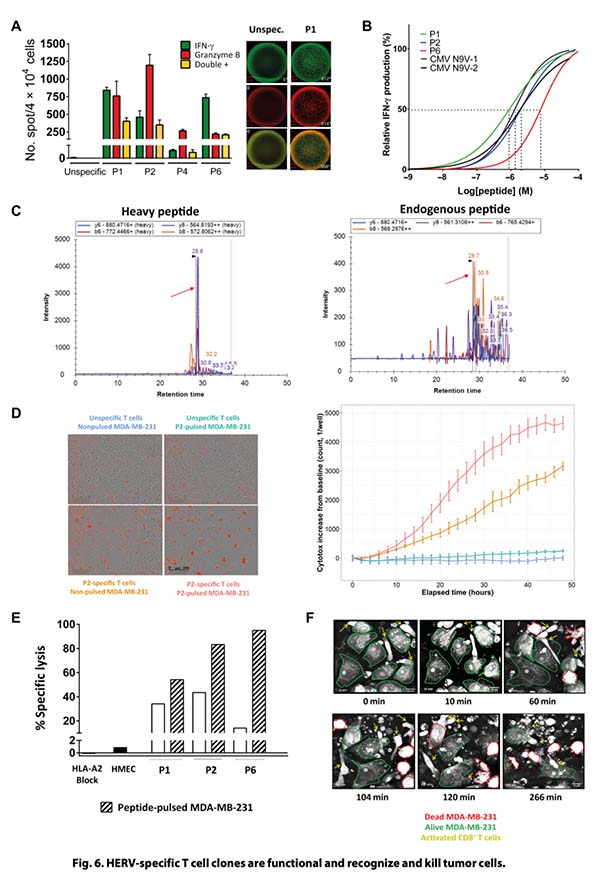
Using a machine learning–guided discovery strategy, the study identified a focused set of HLA-A2–restricted epitopes derived from tumor-associated HERVs that are selectively expressed across multiple solid tumors, particularly breast cancer. These epitopes were shown to induce high-avidity, polyfunctional cytotoxic T cells capable of recognizing and killing tumor cells as well as patient-derived organoids.
Critically, the breakthrough depended on the ability to directly validate and quantify extremely low-abundance peptide–HLA complexes on tumor cells. This is where Valid-NEO® was essential. Through its groundbreaking sensitivity and clinical-grade reproducibility, Valid-NEO enabled direct confirmation that these predicted HERV epitopes were not merely theoretical computational hits, but genuinely processed, presented, and therapeutically actionable targets. Without this level of sensitivity and reproducibility, many of these low-copy-number antigens would likely have remained invisible to conventional immunopeptidomics approaches.
The study further showed that these HERV-specific T cells are naturally present in tumor-infiltrating lymphocytes and can mediate potent, HLA-restricted tumor killing. Together, the data establish shared HERV epitopes as a compelling new immunotherapy target class, especially for tumors with low or intermediate mutational burden, where classic neoantigen strategies are often limited.
Strategic Significance:
This work validates a major strategic thesis behind Valid-NEO®: the future of cancer immunotherapy will not be limited to personalized mutation targets alone. With the right detection platform, previously hidden, low-abundance peptide–HLA targets can be converted into repeatable, druggable, and potentially scalable immunotherapy opportunities.
More importantly, this study shows that Valid-NEO does not just detect and validate known targets — it enables the discovery of entirely new target classes. By combining ultra-sensitive detection with clinically reproducible validation, Complete Omics is helping expand the addressable immunotherapy universe beyond conventional neoantigens into shared intracellular antigens with real translational and commercial potential.

