A neoantigen from the most frequently mutated tumor suppressor gene in human cancers, TP53, is identified by Complete Omics Inc.
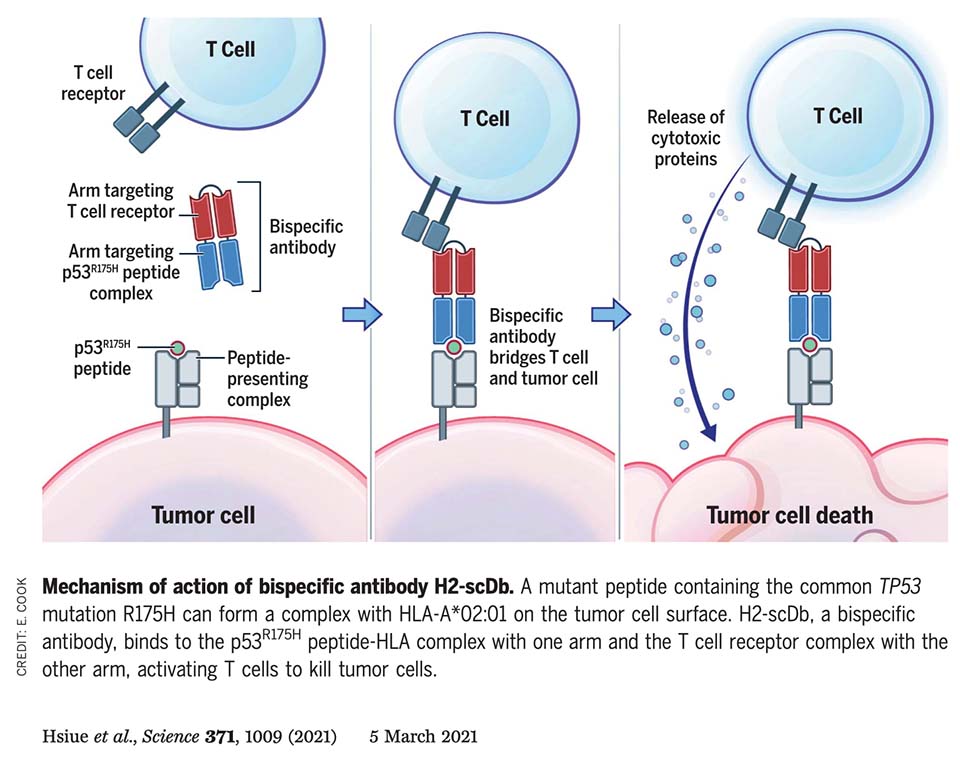
This landmark study demonstrates the power of our Valid-NEO® platform to transform previously “undruggable” cancer mutations into actionable therapeutic targets. The work focuses on TP53 R175H, one of the most common driver mutations in human cancer. Although mutant p53 is an intracellular protein and has historically been impossible to target with conventional drugs, the study shows that fragments of the mutant protein are naturally presented on tumor cells as neoantigen peptide–HLA complexes.
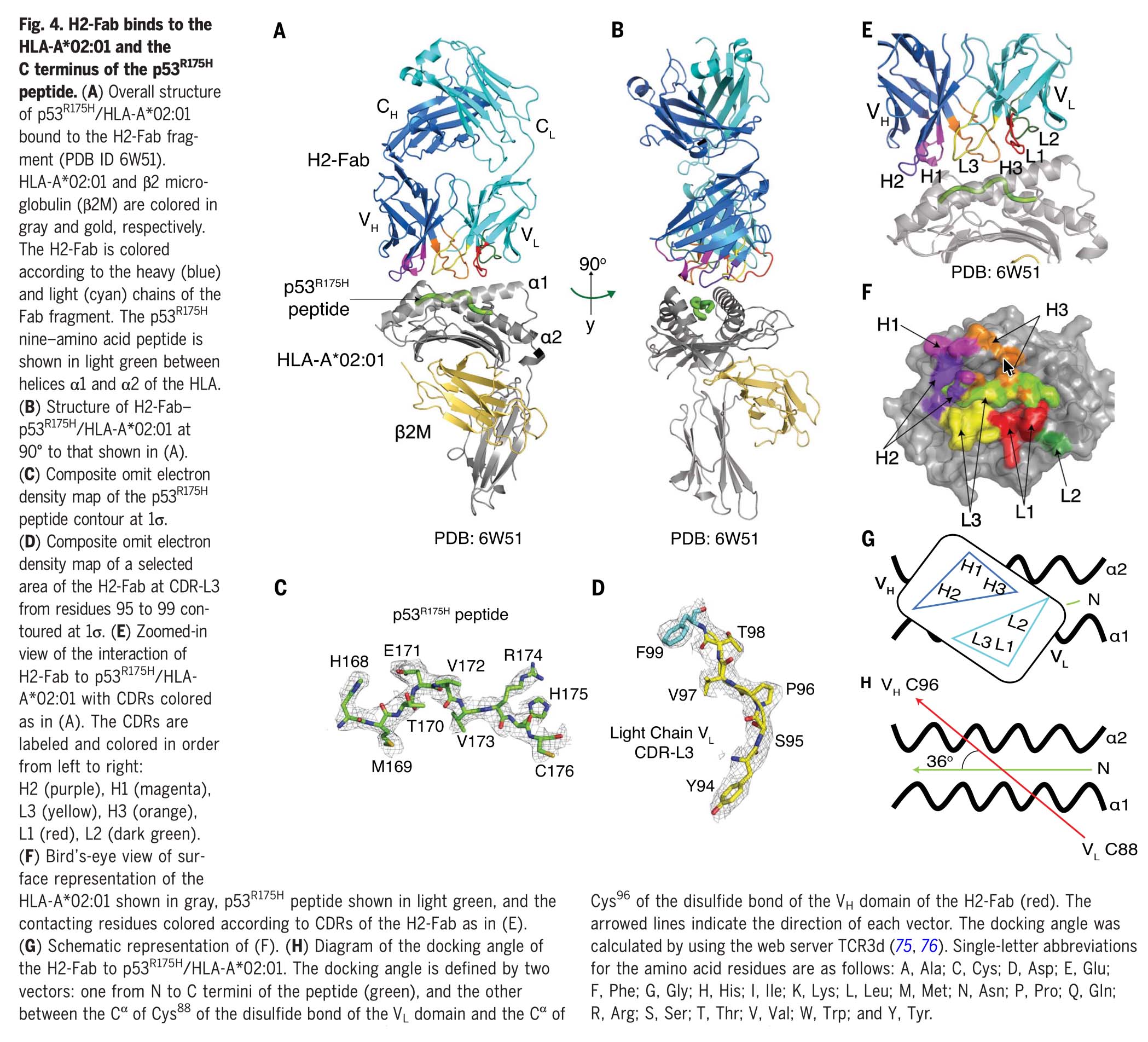
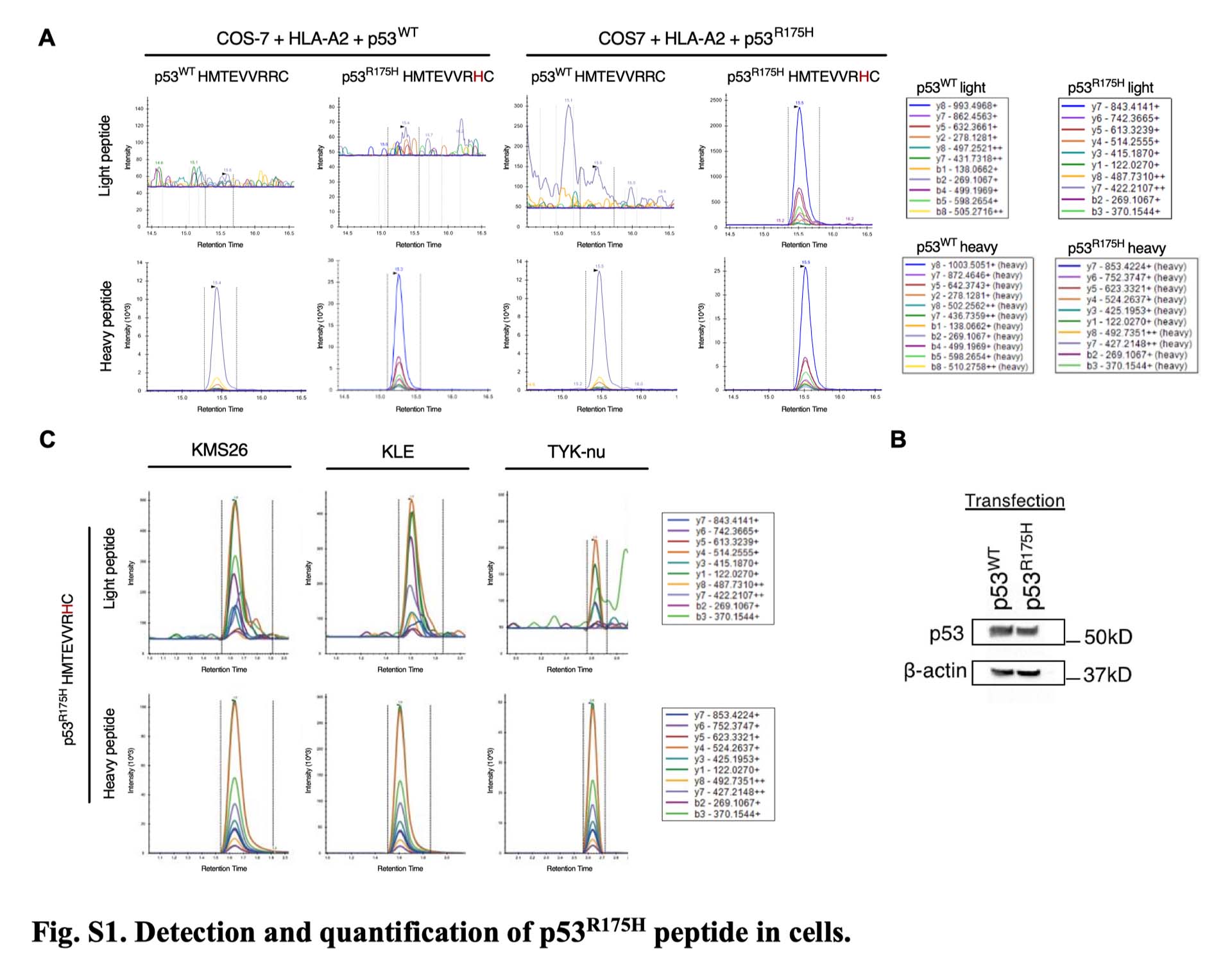
Using high-efficiency immunopeptidomics and targeted validation approaches—the core capabilities underlying Valid-NEO—the team directly detected and quantified this neoantigen on cancer cells. Despite being present at extremely low copy numbers, the neoantigen could be selectively recognized by a highly specific antibody engineered to bind the mutant peptide–HLA complex while ignoring the wild-type protein. The antibody was converted into a bispecific T-cell engager, enabling immune cells to recognize and kill tumor cells harboring the TP53 mutation. The therapy showed potent tumor-killing activity in vitro and significant tumor regression in vivo.
Strategic Significance:
This work validates the central thesis behind Valid-NEO: with the right detection and validation technologies, intracellular driver mutations—long considered inaccessible—can be converted into precise, targetable cancer immunotherapies. This capability unlocks a vast new category of therapeutic targets across oncology.

