HERV-Derived Shared Tumor Epitopes in Ovarian Cancer Identified with Valid-NEO®
A new class of shared immunotherapy targets in ovarian cancer was discovered and functionally validated using Complete Omics’ Valid-NEO® ultra-sensitive immunopeptidomics platform.
Shared HERV-derived targets for ovarian cancer immunotherapy
This study extends the HERV immunotherapy thesis into ovarian cancer, one of the most difficult solid tumors for immuno-oncology and a disease where checkpoint inhibitors have shown limited efficacy. The work demonstrates that HERV-K-derived epitopes are selectively expressed and presented by ovarian tumor cells, while remaining undetectable in normal cells from critical tissues.
Using integrated transcriptomics, immunohistochemistry, targeted immunopeptidomics, and functional T-cell assays, the study showed that these shared HERV-derived epitopes can induce high-avidity cytotoxic CD8+ T cells that specifically recognize and kill ovarian cancer cells in vitro and in vivo. Importantly, HERV-specific T cells were also detected in tumor-infiltrating lymphocytes and could be generated from patients with ovarian cancer, establishing both biological relevance and translational feasibility.
Unlike patient-specific mutation-derived neoantigens, these HERV-derived targets represent a shared antigen class, creating the possibility of scalable vaccines, TCR-based cell therapies, and T-cell-engaging biologics for broader patient populations.
Valid-NEO® made these targets experimentally actionable
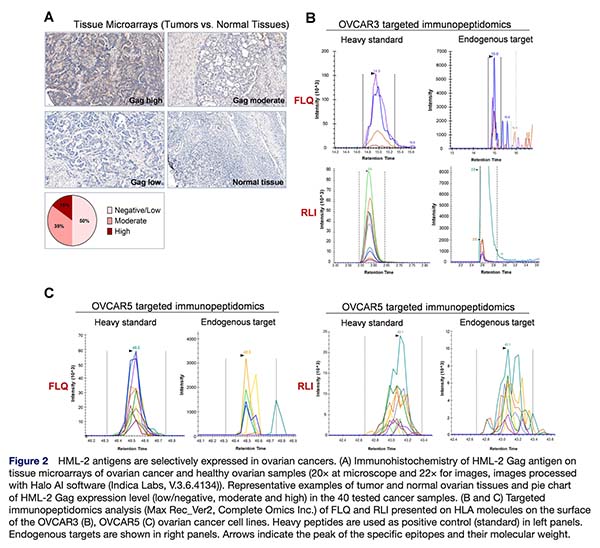
The central technical challenge in this program was proving that the predicted HERV-derived peptides are actually presented on HLA molecules on the tumor cell surface at levels relevant for immune targeting.
Complete Omics’ Valid-NEO® platform was the enabling technology that made this possible. Through its exceptional sensitivity, optimized peptide-HLA enrichment, MaxRec sample-preservation workflow, and clinically reproducible targeted detection, Valid-NEO directly confirmed the presence of HERV-derived epitopes on ovarian cancer cell lines while showing no detectable presentation on tested normal primary cells from critical tissues.
This is a critical distinction. Many candidate tumor antigens remain trapped at the computational or transcriptomic level. Valid-NEO converts prediction into proof by demonstrating true peptide presentation, the key step required to transform a theoretical target into a therapeutically actionable one. In this study, that capability was essential for de-risking the target class and validating tumor selectivity.
Strong preclinical evidence for multiple therapeutic formats
The study generated compelling preclinical support for HERV-targeted immunotherapy in ovarian cancer. HERV-specific T cells displayed high functional avidity, killed multiple ovarian cancer cell lines in an HLA-restricted manner, spared normal primary cells, and showed significant antitumor activity in an in vivo avian embryo model, reducing tumor volume by approximately 63% versus controls.
These findings support multiple downstream product strategies, including:
- Off-the-shelf cancer vaccines
- Adoptive T-cell therapies / TCR-T
- T-cell-recruiting bispecific antibodies
This creates a broad platform opportunity around a shared antigen class in a setting where current immunotherapy options remain inadequate.
Strategic significance
This work reinforces a core Complete Omics thesis: the next wave of immuno-oncology value creation will come from previously hidden, low-abundance, but shared tumor antigens that conventional technologies cannot reliably validate.
In ovarian cancer, where mutational burden is relatively low and personalized neoantigen approaches are harder to scale, HERV-derived epitopes offer a compelling alternative. Valid-NEO® was the critical platform that enabled these targets to be seen, confirmed, and advanced toward therapeutic development.
This is not just a biomarker story. It is a target discovery and target validation story with direct implications for drug development. By enabling precise detection of rare peptide-HLA complexes with clinical-grade reproducibility, Valid-NEO expands the immunotherapy target universe and helps turn difficult cancers into actionable markets.

