Shared Graft-Versus-Leukemia Minor Histocompatibility Antigens Discovered with Valid-NEO®
A large set of novel shared minor histocompatibility antigens (mHAs) driving graft-versus-leukemia (GVL) responses was discovered and experimentally validated using Complete Omics’ Valid-NEO® ultra-sensitive immunopeptidomics platform.
Expanding the therapeutic target space for graft-versus-leukemia immunotherapy
Allogeneic hematopoietic cell transplantation (alloHCT) remains one of the most effective curative treatments for high-risk acute myeloid leukemia (AML), reducing relapse risk by more than 60% compared with chemotherapy alone. The therapeutic benefit arises from donor T-cell recognition of minor histocompatibility antigens (mHAs) expressed by leukemia cells. However, this immune response presents a long-standing clinical challenge: the same T-cell responses that eliminate leukemia cells can also attack healthy tissues, causing graft-versus-host disease (GVHD). A key strategy to separate beneficial graft-versus-leukemia (GVL) activity from harmful GVHD is to target mHAs selectively expressed in hematopoietic tissues.
Historically, discovery of such targets has been extremely limited. Prior to this work, only ~12 validated class I GVL mHAs had been identified over several decades. This study dramatically expands that landscape. Using a computational pipeline integrating SNP mismatch analysis, tissue-expression filtering, and HLA-binding prediction across 3,231 donor-recipient pairs from the DISCOVeRY-BMT cohort, researchers predicted nearly 1.9 million potential GVL mHAs and identified highly shared candidates across common HLA alleles. Importantly, the analysis revealed that although most mHAs are patient-specific, a small subset are widely shared across transplant recipients, creating the possibility of off-the-shelf immunotherapies targeting leukemia-specific antigens.
Valid-NEO® enabled direct experimental confirmation of shared mHAs
A critical step in translating computational predictions into therapeutic targets is confirming that candidate peptides are actually presented on HLA molecules on leukemia cells.
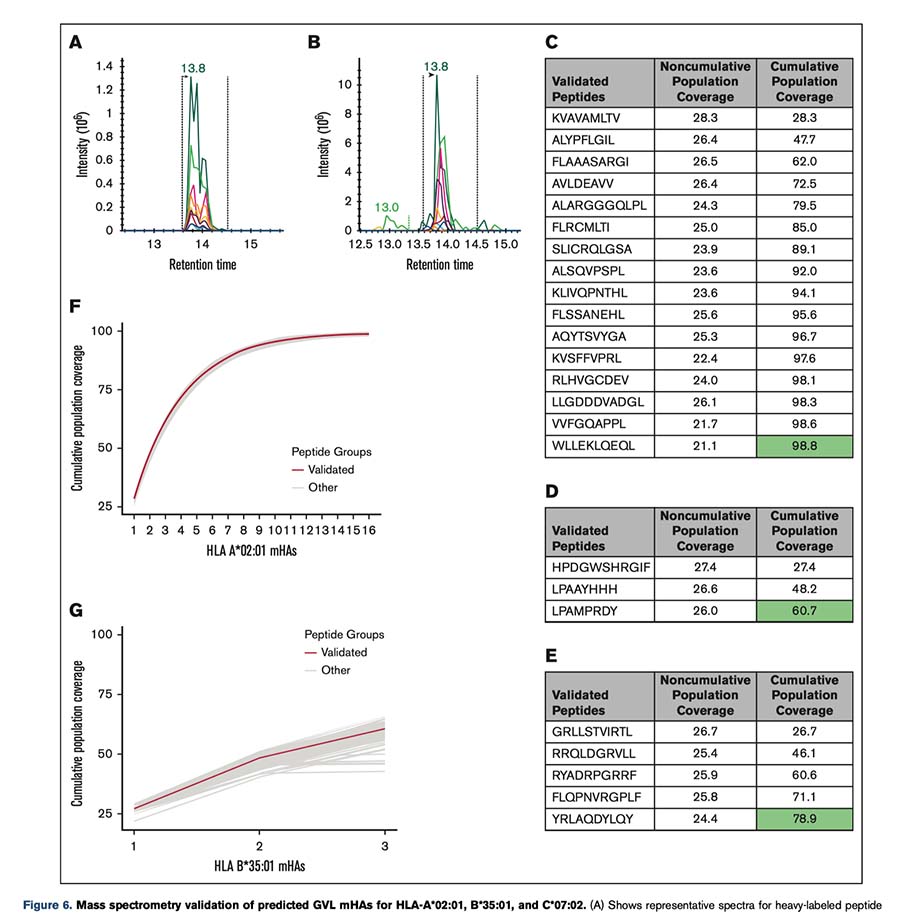
This validation step was performed using Complete Omics’ Valid-NEO® platform, which enables highly sensitive enrichment and targeted mass-spectrometry detection of peptide–HLA complexes.
Using Valid-NEO immunopeptidomics, the team screened predicted targets in leukemia cell lines and successfully validated 24 previously unknown GVL minor histocompatibility antigens presented on HLA class I molecules:
- 16 mHAs presented by HLA-A*02:01
- 3 mHAs presented by HLA-B*35:01
- 5 mHAs presented by HLA-C*07:02
These validated targets collectively cover a substantial proportion of transplant recipients in the DISCOVeRY-BMT dataset:
- 98.8% of HLA-A*02:01 patients
- 60.7% of HLA-B*35:01 patients
- 78.9% of HLA-C*07:02 patients
The study also demonstrated functional immunogenicity, showing that one of the newly identified antigens (UNC-HEXDC-V) can stimulate antigen-specific CD8⁺ T-cell responses.
By enabling direct detection of these peptide-HLA complexes, Valid-NEO converted predicted targets into experimentally validated immunotherapy candidates.
Strategic significance
This work increases the number of known validated class I graft-versus-leukemia minor histocompatibility antigens by approximately 200%, representing one of the largest expansions of the mHA target landscape since the first discovery of HA-1 in 1983.
More importantly, the study demonstrates that shared mHAs exist at clinically meaningful population frequencies, enabling the possibility of scalable antigen-directed therapies after transplantation, including:
- TCR-engineered T-cell therapies
- peptide or RNA vaccines
- donor lymphocyte infusion strategies
- bispecific T-cell-engaging biologics
From a platform perspective, this work highlights the importance of ultra-sensitive immunopeptidomics for validating rare peptide-HLA complexes that cannot be confirmed using conventional discovery methods.
By transforming predicted genomic mismatches into validated immune targets, Valid-NEO expands the therapeutic antigen space for hematologic malignancies and supports the development of next-generation GVL-specific immunotherapies that preserve anti-leukemia efficacy while minimizing GVHD risk.

